Effects of cold stress on leaf physiological characteristics in Forsythia suspensa (Thunb.) Vahl seedlings
-
摘要:
连翘(Forsythia suspensa (Thunb.) Vahl)作为常见的早春花卉和大宗药用作物,低温是其在春季面临的主要胁迫因素。本研究以连翘幼苗叶片为试材,测定其在4个低温条件下的相对电导率、丙二醛(MDA)、脯氨酸(Pro)、可溶性糖(SS)、可溶性蛋白(SP)和叶绿素含量、超氧化物歧化酶(SOD)和过氧化物酶(POD)活性以及叶绿素荧光参数等指标的变化。结果显示,低温胁迫引起连翘叶片相对电导率和MDA含量升高。连翘叶片通过多种途径抵御低温伤害,SP、SS、SOD在胁迫前期发挥主要作用,胁迫增强时Pro和POD发挥更为主要的作用。JIP-test分析结果表明,低温对光合作用产生显著影响,主要体现在对电子传递的抑制和受体侧的伤害上,0℃处理伤害最为严重。低温胁迫影响连翘幼苗生长,4℃以上低温胁迫解除后,其生长可得到恢复,而0℃低温胁迫对其造成的伤害不可逆。
Abstract:Forsythia suspensa (Thunb.) Vahl is a common early spring flower and bulk cultivated medicinal crop. Low temperature is the main stress factor for this species in spring. To date, however, no physiological research has explored the effects of low temperature on F. suspensa growth. Here, we used the leaves of F. suspensa seedlings and detected changes in relative conductivity, malondialdehyde (MDA), proline content (Pro), soluble sugar (SS), soluble protein (SP), chlorophyll content, superoxide dismutase (SOD), peroxidase (POD) activity, and chlorophyll fluorescence parameters under four temperature conditions. We also detected chlorophyll fluorescence parameters after recovery. Results showed that low temperature stress caused an increase in relative electrical conductivity and MDA content in the leaves, which affected growth to some extent. The resistance of F. suspensa leaves to low temperature was achieved by multiple pathways. SP, SS, and SOD played major roles at the early stage of cold stress, while Pro and POD played important roles when stress intensity increased. The JIP-test showed that low temperature had a significant effect on photosynthesis, mainly reflected in the inhibition of electron transfer and damage to the receptor side. Treatment damage at 0℃ was the most serious. Low temperature stress affected the growth of F. suspensa seedlings. Low temperature stress above 4℃ affected the seedlings, but growth recovered after the stress was removed. Damage caused by low temperature stress at 0℃ was irreversible. Thus, F. suspensa should be cultivated in areas where low temperatures are less common in late spring, or cold-tolerant varieties should be selected for cultivation in areas where low temperatures are more frequent in late spring.
-
Keywords:
- Forsythia suspense /
- Cold stress /
- Photosynthesis /
- JIP-test /
- Chlorophyll fluorescence
-
低温胁迫是影响植物生长发育的重要因素之一。在低温下,植物的生理代谢和分子过程会受到不同程度的影响[1]。连翘(Forsythia suspensa (Thunb.) Vahl为木犀科连翘属多年生木本植物。作为常见的早春花卉和大宗药用作物,其面临的主要胁迫因素就是春季低温。尤其近几年来气候波动较大,造成连翘幼苗成活率低、产量下降[2]。因此,开展连翘低温胁迫生理响应相关研究对理解连翘抗寒机制和抗寒品种培育具有重要意义。
生理指标变化是植物受到冷害表型发生变化的前期反应,指示低温对植物影响的生理指标有三类:一是反映植物受低温胁迫伤害程度的指标,如电导率、丙二醛(MDA)等。当温度低于临界值时,植物的膜系统会最先受到伤害,其透性增加,水溶性物质外渗,导致相对电导率增加;当胁迫进一步加剧,膜脂会发生降解,从而导致植物死亡,电导率越高表示伤害越大[3];低温胁迫还可造成植物产生过量活性氧(ROS),细胞膜脂被过氧化会生成MDA,其含量的高低反映了细胞膜脂过氧化水平[4]。二是植物产生保护性反应的生理指标,如超氧化物歧化酶(SOD)、过氧化物酶(POD)等。酶能清除低温胁迫下产生的过量ROS,有效降低膜脂过氧化,所以酶活性的变化反映了植物对低温胁迫的响应[5],不同物种可能利用不同的保护酶体系应对低温胁迫[6, 7]。此外,低温胁迫后,渗透调节物如可溶性蛋白(SP)、可溶性糖(SS)、脯氨酸(Pro)等含量升高,从而平衡植物细胞的渗透压,降低或消除低温造成的伤害[8]。蛋白质代谢对生物生长发育至关重要,同时也是植物应对低温胁迫的一个重要方面[9]。SS和Pro含量提升可提高细胞液浓度,增加细胞的保水能力,增强细胞液的流动性,维持细胞膜在低温下的正常功能[10],它们也是响应低温胁迫的重要生理指标。三是与植物光合作用相关的指标,光合器官是植物对低温最敏感的部位之一,低温胁迫会增加其膜的粘度,并限制质体醌(PQ)的扩散和抑制类囊体的电子传输[11]。绿色植物存在光系统Ⅰ(PSⅠ)和光系统Ⅱ(PSⅡ),PSⅠ和PSⅡ天线捕获光能及其在反应中心(RC)驱动电荷分离时很容易受到低温干扰,尤其是PSⅡ对低温胁迫更为敏感[12]。目前常通过分析PSⅡ光系统变化来研究植物对低温胁迫的响应[13]。以往对连翘的研究主要关注其药理[14]和化学成分[15],有关低温对连翘生长影响的报道并不多。本文通过测定不同低温处理下的相对电导率、MDA、SP、SS和Pro含量、SOD和POD活性、叶绿素荧光参数等的变化,旨在理解连翘抗寒的生理机制,并为抗寒品种的培育提供理论基础。
1. 材料与方法
1.1 材料
连翘种子由三门峡大美连翘有限公司提供,挑选300粒颗粒饱满的种子置于0.5%的高锰酸钾水溶液中浸种2 h,用蒸馏水充分冲洗,之后置于蒸馏水中浸种催芽8 h。将催过芽的连翘种子播种于穴盘中,选用混合基质(河沙∶珍珠岩∶草炭土 = 1∶2∶3)进行培养,培养温度为25℃ / 20℃(昼14 h / 夜10 h),光照5000 lx,湿度60%。待连翘幼苗长出2~4片真叶时移栽至营养钵(9 cm × 11 cm),每盆1株,用相同基质继续培养,培养温度为28℃ / 20℃(昼14 h / 夜10 h),光照15000 lx,湿度60%。育苗实验在河南农业大学风景园林与艺术学院公共实验创新平台人工气候箱(PLD-500-G4,宁波乐电仪器)中进行。待幼苗长至平均株高为(20 ± 5)cm、叶片数为(16 ± 3)片时,选取外部形态和长势基本一致且健壮、无病虫害的连翘幼苗进行低温处理。
1.2 材料处理
实验共设4个温度处理:25℃(T1)、8℃(T2)、4℃(T3)、0℃(T4),将材料分别置于4个人工气候箱处理36 h,培养条件为光照5000 lx,湿度60%,以25℃处理为对照。处理结束后选取自上向下数第3~6片健康的功能叶进行叶绿素荧光瞬态的测定,并做好标记,以便恢复处理(25℃处理24 h)后再次测定。分别用T1-H、T2-H、T3-H、T4-H表示4个温度的常温恢复处理。部分叶片用于电导率测定,其他立即投入液氮冷冻,置于−80℃冰箱保存,用于后续相关生理指标测定。
1.3 生理指标的测定
MDA含量采用硫代巴比妥酸法[16]测定、SOD和POD活性、SP含量测定均参考王永军等[17]的方法。相对电导率采用Redmann等[18]的方法测定,叶绿素含量采用95%乙醇浸提法[19],SS含量测定采用Rosa等[20]的方法,Pro含量测定采用Bates等[21]的方法,每个处理测定3个生物学重复。
1.4 叶绿素荧光参数的测定
室温下,利用多功能植物效率分析仪M-PEA(Hansatech Instruments公司,英国)测定连翘幼苗叶片相同部位的叶绿素荧光瞬态。测定时选取并标记从顶芽向下的第3、4片健康叶子,测定前进行30 min的暗适应处理,以确保其初始光化学反应为0,测定时光脉冲强度为3000 μmol·m−2·s−1,持续时间为3 s,每个处理10个生物学重复。
参照JIP-Test测定方法[22,23],利用多相态荧光的原始数据对叶绿素荧光进行分析,测定相关叶绿素荧光参数值,相关参数及其意义见附表1
1 。1.5 数据处理
利用Excel 2010和SPSS 19.0软件进行数据统计和差异分析,使用Excel 2010和Origin 9.1软件绘制图表。
2. 结果与分析
2.1 低温胁迫对连翘幼苗叶片各生理指标的影响
与对照相比,各指标随着温度降低均表现出明显的变化(表1)。随着胁迫程度加剧,连翘幼苗叶片的相对电导率和MDA含量均呈升高趋势,T3和T4组的相对电导率分别比T1高0.87和2.39倍, 而T4组连翘叶片MDA含量比T1高1.27倍,表明连翘幼苗在0℃低温下叶片膜脂过氧化严重。
表 1 不同温度下连翘幼苗叶片各生理指标的变化Table 1. Changes in physiological indices in leaves of Forsythia suspensa seedlings at different temperatures生理指标
Physiological index处理 Treatment T1 T2 T3 T4 相对电导率 0.230 ± 0.01a 0.290 ± 0.01ab 0.430 ± 0.02b 0.580 ± 0.01c MDA含量 /
μmol/g FW0.015 ± 0.00a 0.025 ± 0.00b 0.028 ± 0.00b 0.034 ± 0.00c SP含量 /
mg/g FW4.180 ± 0.04b 4.471 ± 0.04c 4.516 ± 0.04c 4.359 ± 0.03a SS含量 /
μg/g FW28.883 ± 0.31a 46.577 ± 0.27b 64.277 ± 0.19c 59.265 ± 0.19c Pro含量 /
μg/g FW22.794 ± 0.12a 19.750 ± 0.03a 35.925 ± 0.17b 40.482 ± 0.35b 叶绿素a /
mg/g FW0.430 ± 0.00a 0.449 ± 0.00ab 0.443 ± 0.00a 0.445 ± 0.00ab 叶绿素 b /
mg/g FW0.808 ± 0.00b 0.584 ± 0.02a 0.623 ± 0.02a 0.585 ± 0.02a 叶绿素总含量 /
mg/g FW1.238 ± 0.00b 1.033 ± 0.01a 1.066 ± 0.02a 1.030 ± 0.02a SOD活性 /
U/g FW419.542 ± 10.02a 431.441 ± 6.87ab 479.531 ± 2.48c 441.430 ± 5.05b POD活性 /
U/g FW11.725 ± 1.10a 15.535 ± 1.63a 25.331 ± 2.69b 27.879 ± 1.26b 注:同行不同小写字母表示处理间差异显著(P < 0.05)。 Note: Different lowercase letters indicate significant differences between treatments (P < 0.05). SP和SS随着胁迫增加均呈先升高后降低的变化趋势,在T3组达到最大值,分别比T1高0.08和1.23倍,在T4组两者含量有所下降,但仍高于T1,其中SS与T1相比差异显著。连翘叶片的Pro含量则随温度降低先缓慢下降,然后急剧上升,在T4组达到最大值,比对照高0.78倍。
总叶绿素和叶绿素b含量随胁迫加重均呈明显的下降趋势,而叶绿素a的变化不显著。低温处理下连翘幼苗叶片中SOD活性呈先上升后下降趋势,在T3组达最大值,随着胁迫强度的进一步提升,其活性开始降低。POD活性随温度降低呈显著上升趋势,T4组比T1组高1.38倍。
2.2 低温胁迫对连翘叶片快速叶绿素荧光的影响
2.2.1 低温胁迫下OJIP曲线变化
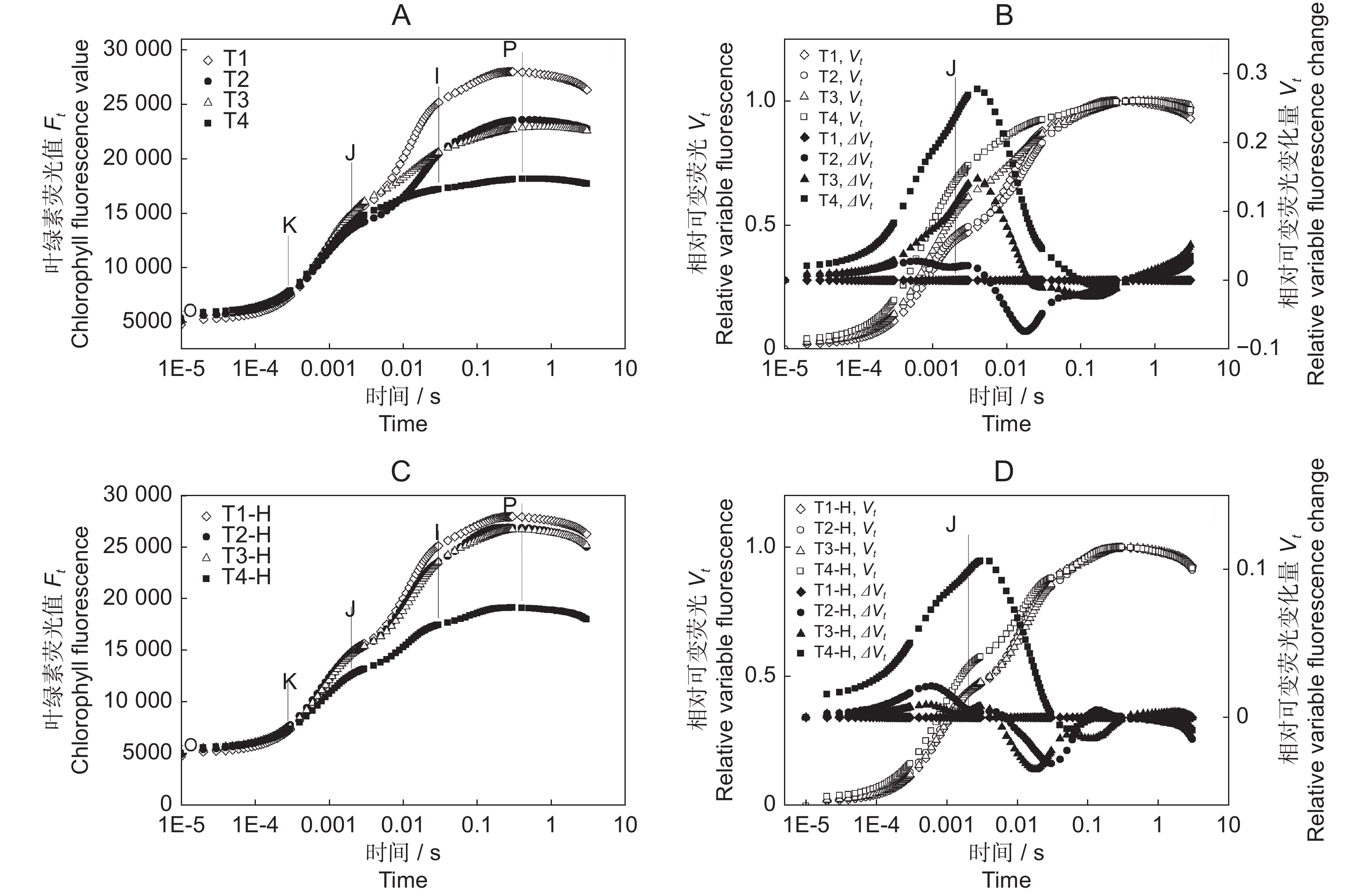
从O点到P点的荧光上升过程称为快速叶绿素荧光诱导动力学曲线,主要反映了PSⅡ的原初光化学反应及光合机构电子传递状态等过程的变化。O、J、I、P 4相随温度降低以及恢复处理后的变化趋势如图1:A和1:C所示,T2和T3组的变化趋势相似,T4组变化最显著;恢复处理后,除T4组幼苗未恢复,其他组均接近对照水平,表明0℃低温处理对光合系统的伤害最严重。Vt是荧光值Ft通过(Fm−FO)标准化得到,而ΔVt = Vt处理−Vt对照,图1:B和图1:D分别显示了低温处理和恢复处理连翘幼苗与对照的OJIP曲线之差,曲线均在J点附近出现清晰的峰值。
![]() 图 1 低温胁迫及恢复后连翘叶片快速叶绿素荧光变化A:低温处理后快速叶绿素荧光曲线;B:低温处理后相对可变荧光曲线;C:恢复后快速叶绿素荧光曲线;D:恢复后相对可变荧光曲线。Figure 1. Rapid chlorophyll fluorescence changes in Forsythia suspensa seedlings under cold stress and recoveryA: Fast chlorophyll fluorescence curve after cold stress; B: Relative variable fluorescence curve after cold stress; C: Fast chlorophyll fluorescence curve after recovery; D: Relative variable fluorescence curve after recovery.
图 1 低温胁迫及恢复后连翘叶片快速叶绿素荧光变化A:低温处理后快速叶绿素荧光曲线;B:低温处理后相对可变荧光曲线;C:恢复后快速叶绿素荧光曲线;D:恢复后相对可变荧光曲线。Figure 1. Rapid chlorophyll fluorescence changes in Forsythia suspensa seedlings under cold stress and recoveryA: Fast chlorophyll fluorescence curve after cold stress; B: Relative variable fluorescence curve after cold stress; C: Fast chlorophyll fluorescence curve after recovery; D: Relative variable fluorescence curve after recovery.快速叶绿素荧光测定前将叶片暗处理30 min,此时,反应中心会完全打开,且其相关的醌被完全氧化。光照开始时只有一小部分能量消散(FO);相反,当所有反应中心均关闭且醌被完全还原时,荧光将达到最大值(Fm)。低温胁迫以及恢复后的FO均随温度降低呈上升趋势。最大荧光Fm呈下降趋势,T2和T3组在恢复后逐渐升高,而T4组经常温恢复处理后,其Fm并没有得到明显恢复(附表2
2 )。2.2.2 低温胁迫下连翘叶片单位横截面积比活性参数变化
随着低温胁迫程度的加剧,叶片单位面积吸收(ABS/CSO)和耗散的光能(DIO/CSO)显著提升,在T4组达到最高值,分别是T1组的1.13和1.98倍。温度恢复后,ABS/CSO开始下降至接近对照水平,DIO /CSO在T2-H和T3-H组也有所降低,但在T4-H组仍然显著上升。叶片单位面积捕获的光能(TRO /CSO)随温度降低变化不大,但量子产额(ETO /CSO)显著降低,温度恢复后有所提升(表2)。
表 2 低温胁迫及恢复后叶片单位横截面积比活性参数变化Table 2. Changes in specific activity parameters per unit cross-sectional area of leaves after low temperature stress and recovery处理
TreatmentABS/CSO DIO/CSO TRO/CSO ETO/CSO T1 5085.889 ± 126.80a 926.271 ± 34.89a 4159.618 ± 93.10ab 2367.616 ± 45.67c T2 5451.889 ± 72.15ab 1309.842 ± 96.39b 4142.047 ± 103.44ab 2297.065 ± 116.57c T3 5615.000 ± 96.28b 1383.823 ± 50.81b 4231.177 ± 77.20b 1937.618 ± 134.92b T4 5728.333 ± 266.44b 1833.883 ± 153.36c 3894.450 ± 131.31a 1291.324 ± 135.30a T1-H 5099.666 ± 126.60a 920.641 ± 33.09a 4239.601 ± 91.10b 2398.331 ± 44.77b T2-H 5317.111 ± 125.08a 1052.801 ± 38.45a 4264.310 ± 92.68b 2412.295 ± 100.46b T3-H 5429.833 ± 172.32a 1106.154 ± 56.32a 4323.679 ± 119.74b 2453.114 ± 84.32b T4-H 5513.555 ± 262.21a 2072.676 ± 465.71b 3440.880 ± 390.15a 1491.727 ± 270.88a 注:同列不同小写字母表示处理间差异显著(P < 0.05)。下同。 Note: Different lowercase letters in the same column indicate significant difference between treatments (P < 0.05). Same below. 2.2.3 低温对连翘叶片单位PSⅡ反应中心比活性参数的影响
低温胁迫下,QA处在可还原态时,ABS / RC和TRO / RC变化均不明显, ETO / RC呈先升后降的变化趋势,造成光能耗散(DIO / RC)的显著增加,与对照相比,DIO / RC整体升高,T4是对照T1组的2.35倍(表3)。温度恢复后,TRO / RC、ETO / RC可恢复到对照水平,但单位PSⅡ反应中心对光能的吸收(ABS/RC)和耗散(DIO / RC)在T2-H和T3-H组仍有所下降,但T4-H组反而显著提升。
表 3 低温胁迫及恢复后连翘幼苗叶片单位PSⅡ反应中心比活性参数变化Table 3. Changes in specific activity parameters of unit PSII reaction center of Forsythia suspensa seedlings after cold stress and recovery处理
TreatmentABS / RC TRO / RC DIO / RC ETO / RC T1 0.959 ± 0.04a 0.784 ± 0.03a 0.175 ± 0.01a 0.445 ± 0.01b T2 1.218 ± 0.06b 0.918 ± 0.03b 0.300 ± 0.04b 0.512 ± 0.03b T3 1.017 ± 0.03a 0.765 ± 0.02a 0.252 ± 0.02ab 0.346 ± 0.02a T4 1.275 ± 0.09b 0.863 ± 0.05ab 0.412 ± 0.04c 0.280 ± 0.03a T1-H 0.950 ± 0.04a 0.774 ± 0.03a 0.175 ± 0.01a 0.442 ± 0.01b T2-H 1.130 ± 0.03a 0.906 ± 0.02a 0.224 ± 0.01ab 0.512 ± 0.02b T3-H 1.037 ± 0.04a 0.826 ± 0.03a 0.211 ± 0.01ab 0.467 ± 0.01b T4-H 1.804 ± 0.41b 0.895 ± 0.07a 0.910 ± 0.42b 0.360 ± 0.03a 2.2.4 低温胁迫下光系统供体侧和受体侧的变化
当PSⅠ和PSⅡ供体侧受到伤害时,叶绿素荧光在照光后大约300 µs处强度就会上升,出现K相。Wk能反映PSⅡ供体侧受到的伤害。低温处理后Wk随温度变化不明显,温度恢复后也没有发生显著改变(表4),表明低温对连翘叶片PSⅡ供体侧没有造成太大伤害。
表 4 低温胁迫及恢复后连翘叶片供体侧和受体侧参数的变化Table 4. Changes in donor side and receptor side parameters of Forsythia suspensa seedlings after cold stress and recovery处理
TreatmentWk Sm N ψO φEO T1 0.259 ± 0.01a 15.663 ± 0.50a 12.160 ± 0.18a 0.571 ± 0.01c 0.467 ± 0.01c T2 0.310 ± 0.01ab 21.786 ± 1.47b 19.921 ± 1.43b 0.555 ± 0.02c 0.421 ± 0.02c T3 0.259 ± 0.00a 22.336 ± 3.11b 16.767 ± 1.97b 0.459 ± 0.03b 0.347 ± 0.03b T4 0.292 ± 0.01ab 12.877 ± 1.63a 10.897 ± 1.44a 0.333 ± 0.04a 0.230 ± 0.03a T1-H 0.258 ± 0.01a 15.363 ± 0.50ab 12.150 ± 0.18a 0.571 ± 0.01b 0.467 ± 0.01b T2-H 0.271 ± 0.01ab 16.596 ± 0.55bc 15.077 ± 0.77b 0.565 ± 0.01b 0.453 ± 0.01b T3-H 0.248 ± 0.01a 18.958 ± 0.89c 15.617 ± 0.82b 0.567 ± 0.01b 0.452 ± 0.01b T4-H 0.270 ± 0.01ab 12.948 ± 1.55a 11.173 ± 1.24a 0.435 ± 0.06a 0.286 ± 0.06a PSⅡ受体侧主要包括QA、QB和PQ库等。电子从QA−进入电子传递链越多,到达Fm所需要的时间就越长,Sm的值也越大。随低温胁迫的加剧,Sm呈先升高后降低趋势,T3组达最大值,而T4组显著下降,比T1降低了17.8%。温度恢复处理后,T4-H连翘幼苗Sm较恢复前没有显著变化。N的变化趋势与Sm一致。φEO、ψO均呈现整体下降趋势,T4比T1分别降低了41.6%和51.2%。恢复常温后,ψO、φEO均均有所提升,T2-H和T3-H组均恢复至正常水平,但T4-H组回升缓慢。Sm、N在T4-H组没有明显变化,而ψO、φEO则缓慢回升(表4)。
2.2.5 低温胁迫下最大量子产额、PSⅠ受体侧及性能指数的变化
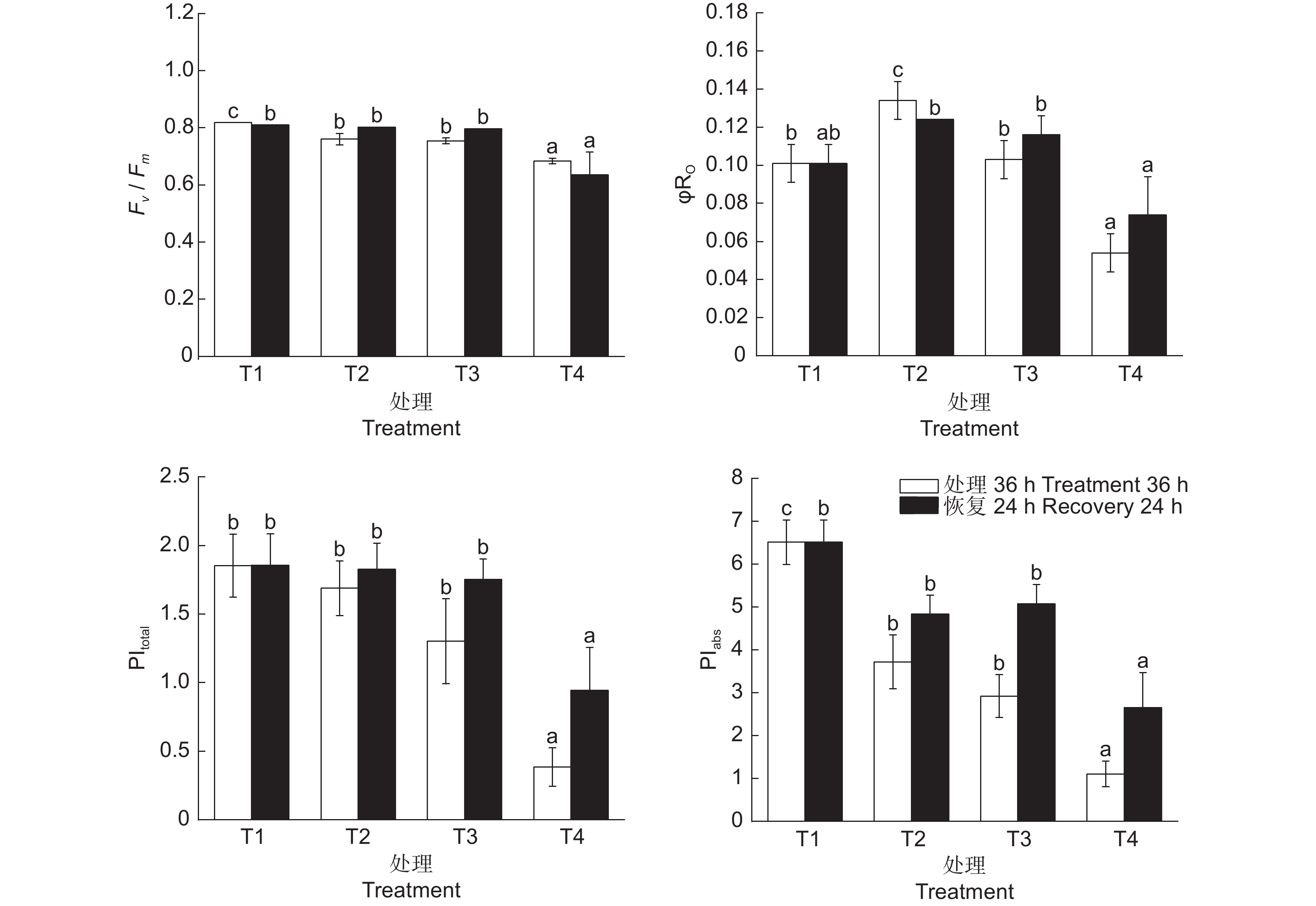
连翘叶片中PSⅡ的最大量子效率(Fv / Fm)在0℃处理时达到最低值,比对照低16.5%,表明低温使连翘对光能的利用率降低(图2)。在各处理中φRO呈先升高后降低的趋势,8℃处理比对照高22.5%,4℃和0℃处理分别比对照低16%和26.7%。随着温度的降低,光合性能指数(PIabs)呈现显著下降趋势,8℃处理比对照降低了42.9%,而0℃处理比4℃下降62.2%,比对照下降了83%。综合性能指数(PItotal)同样呈下降趋势,8℃处理变化不显著,且温度恢复后很快达到到对照水平,4℃、0℃处理降幅较大,比对照分别下降了29.8%和79.2%。
3. 讨论
3.1 低温胁迫对连翘幼苗叶片生理指标的影响
本研究发现随着温度的降低,连翘叶片相对电导率和MDA含量均呈显著上升趋势,表明低温胁迫对连翘叶片细胞膜造成了明显伤害,促使ROS增加,引起细胞膜脂过氧化,细胞透性增大,造成MDA积累,从而伤害植株,其中0℃低温伤害最为明显。这种现象在小麦(Triticum aestivum L.)[4]、鹰嘴豆(Cicer arietinum L.)[24]等植物中也有发现。
植物受到低温胁迫时,会启动生理和生化调节,包括渗透调节物质含量的改变,从而保护自身免受伤害[25]。本研究检测了不同程度低温胁迫下SP、SS、Pro等渗透调节物质的含量,结果发现SP、SS随胁迫加重呈先升高后降低的趋势,在4℃时达到最大值,在0℃时含量有所降低,但仍高于对照。程祥飞等[26]在菊花(Dendranthema morifolium Ramat.)中也发现随着低温胁迫的加剧,SS含量呈现先增加后降低的趋势。SP是植物体内重要的渗透调节物质,低温胁迫时SP含量的增加可提高细胞液浓度,提高植物的抗逆性,但随着胁迫的加剧,其合成受阻,甚至因被水解而导致含量降低,因此呈先增加后降低的趋势;Pro则呈现先降低后上升的趋势。从指标间的变化差异来看,在连翘受较轻低温胁迫时, SP、SS发挥主要作用,其中SS作用最显著,随着胁迫强度增加(0℃处理), Pro发挥更为主要的作用。另一方面,低温胁迫下抗氧化系统活性也发挥重要作用。温度降低,连翘叶片的抗氧化酶保护系统启动,其SOD酶活性升高,但随着胁迫强度加剧,有害物质积累较多,超出了SOD酶的清除能力,迫使其活性下降;而POD活性呈现持续上升趋势,表明其在ROS清除方面发挥了更重要的作用。
低温是限制植物生长发育和地理分布的重要限制因子。为了降低“倒春寒”现象带来的损失,研究低温胁迫对植物的生理影响尤为重要,同时对植抗逆品种的栽植和种植区域的选择具有重要指导意义。连翘野生种群广泛分布于中国暖温带地区[27],在早春2月至3月底容易遭遇低温而影响其生长。李清亚等[28]在对不同豆梨(Pyrus calleryana Decne.)品种的耐寒性进行综合评价时发现,随着低温胁迫加剧,SOD和POD的活性都呈先上升后下降的趋势,而耐冷性强的豆梨品种SOD活性显著高于其他品种,更有利于清除活性氧对细胞膜的危害。耐冷性强的品种可以忍受较强和较长时间的低温胁迫,在实际生产中应该选用耐冷性强的连翘品种进行栽植。近年来,对不同外源物质提高植物抗低温胁迫的研究也引起了特别关注,如Ca2+ [29],脱落酸[30]、水杨酸[31]和油菜素内酯[32]等。这些外源物质的作用机制不同,但都能使植物叶片中Pro含量和过氧化氢酶(CAT)活性增加,从而抵御低温,增加植株耐寒性。露地越冬的连翘幼苗及成苗可施加外源激素以提高其抗寒能力,但外源物质的种类、浓度和效果还需要进一步实验验证。
3.2 低温胁迫对连翘叶片快速叶绿素荧光参数的影响
叶绿素在光合作用中主要参与吸收、传递光能或引起原初光化学反应,其含量直接决定着植物光合作用的强度[33]。在低温胁迫下,连翘叶片叶绿素总含量与对照相比呈下降趋势,其中叶绿素b含量降低较为显著,这可能是低温造成的MDA积累引起了卟啉途径的代谢障碍,从而抑制了叶绿素的合成[34]。
通过JIP测试分析快速叶绿素荧光数据,定量连翘叶片PSⅡ在胁迫条件下的变化[35],能够更清晰地揭示低温对连翘光系统的伤害。本研究中OJIP曲线在J点处出现清晰的峰值,这与QA−的积累有关,即低温可能抑制了QA−的再氧化[36]。另外,随着温度降低,连翘叶片的FO显著提升。初始荧光的强弱与光合色素含量多少及作用中心的活性状态有关,光合色素降解可导致FO的下降;而作用中心失活可导致FO的上升[37],低温可能抑制了连翘叶片PSⅡ作用中心的活性或使其发生了可逆性失活[38]。连翘叶片Fm的变化趋势与FO相反,可能是天线能量耗散(DIO)太大引起的[39]。
低温胁迫后单位受光面积(CSO)的量子效率(ABC / CSO、TRO / CS、ETO / CS和DIO / CS)与单位反应中心的量子效率(ABS / RC、TRO / RC、ETO / RC和DIO / RC)均受到不同影响,电子传递(ETO / CS、ETO / RC)显著降低,而耗散(DIO / CS、DIO / RC)则显著升高。这可能是连翘叶片PSⅡ反应中心响应低温的一种保护机制,低温下植物能吸收光能,但抑制电子传递,待逆境解除后再恢复活性[40]。为了耗散相对较多的剩余激发能,叶片中启动较高的热耗散,以减轻低温造成的伤害。但0℃处理的材料恢复常温后,其对光能的吸收和耗散均显著升高,逆境的解除并没有使其恢复正常。其原因可能是PSⅡ受体侧电子初级受体QA−向二级受体QB的电子传递被抑制,导致电子在QA−处大量积累[33],使光合效率降低且难以恢复,这也与J峰的出现相一致。
叶绿素荧光曲线在300 μs附近是否出现K相可以指示植物放氧复合体是否受到伤害[41]。在低温胁迫过程中,连翘叶片Wk变化并不显著,表明低温对PSⅡ供体侧影响不大。但对PSⅡ受体侧影响较大,8℃和4℃处理的叶片受体侧PQ库大小、QA被还原次数增加,说明从QA−传递的电子较多,即PSⅡ受体侧的电子传递能力相对较高,以此来缓解低温造成的伤害。0℃时φEo、N、受体侧电子传递速度,甚至Sm均呈显著降低趋势,且恢复常温后并没有显著回升,表明0℃时PSⅡ受体侧遭到严重破坏,这与秋季低温对风箱果(Physocarpus amurensis (Maxim.) Maxim.)叶片光系统Ⅱ的影响相似[42],低温对植物光合原初反应造成的伤害主要在PSⅡ反应中心的受体侧。
φRO代表PSⅠ受体侧还原末端电子受体的量子产率[24]。本研究结果表明在8℃和4℃时φRO比对照有所提高,但随后开始降低。由此推测,植物自身能够缓解4℃以上低温导致的PSⅡ损伤,同时电子传递链能够正常运行,使过剩的激发能向PSⅠ转移,减轻因低温导致的伤害,保证光合反应的正常进行。但随着胁迫加剧,PSⅠ和PSⅡ之间平衡被打破,光合作用受到抑制[43]。随着处理温度的降低,连翘的最大量子效率及性能指数均呈降低趋势,表明低温使光合机构受到伤害,降低了光能转化效率。温度恢复后,除了0℃处理外,各处理的光合指标均有所回升,表明光合能力正在恢复。有研究认为,Fv / Fm是体现植物受低温胁迫影响程度的重要指标,胁迫条件下明显下降[44]。本研究中Fv / Fm在低温胁迫下变化显著,但其波动幅度不及PIabs,与本文结果类似,在棉花 (Gossypium hirsutum L.) [45]中也发现PIabs对低温度更敏感。因此,可用PIabs和Fv / Fm共同指示植物对低温的响应程度。
4. 结论
本研究分析了低温胁迫对连翘幼苗叶片生理特性和光合作用的影响,结果发现SP、SS、SOD在轻微胁迫下发挥主要作用,胁迫强度增加后,则是Pro和POD发挥主要作用。低温胁迫影响连翘的生长,但4℃以上低温胁迫后,随着温度的恢复其生长可得到恢复,而0℃低温胁迫对其造成的伤害则不可逆转。光合参数PIabs、Fv / Fm对低温最为敏感,可作为筛选连翘抗寒品种的指标。连翘应选择在较少出现倒春寒的地区种植,或选用耐寒品种在经常出现倒春寒的地区进行栽培。
1 1) 如需查阅附表内容请登录《植物科学学报》网站(http://www.plantscience.cn)查看本期文章。2 1) 如需查阅附表内容请登录《植物科学学报》网站(http://www.plantscience.cn)查看本期文章。 -
图 1 低温胁迫及恢复后连翘叶片快速叶绿素荧光变化
A:低温处理后快速叶绿素荧光曲线;B:低温处理后相对可变荧光曲线;C:恢复后快速叶绿素荧光曲线;D:恢复后相对可变荧光曲线。
Figure 1. Rapid chlorophyll fluorescence changes in Forsythia suspensa seedlings under cold stress and recovery
A: Fast chlorophyll fluorescence curve after cold stress; B: Relative variable fluorescence curve after cold stress; C: Fast chlorophyll fluorescence curve after recovery; D: Relative variable fluorescence curve after recovery.
表 1 不同温度下连翘幼苗叶片各生理指标的变化
Table 1 Changes in physiological indices in leaves of Forsythia suspensa seedlings at different temperatures
生理指标
Physiological index处理 Treatment T1 T2 T3 T4 相对电导率 0.230 ± 0.01a 0.290 ± 0.01ab 0.430 ± 0.02b 0.580 ± 0.01c MDA含量 /
μmol/g FW0.015 ± 0.00a 0.025 ± 0.00b 0.028 ± 0.00b 0.034 ± 0.00c SP含量 /
mg/g FW4.180 ± 0.04b 4.471 ± 0.04c 4.516 ± 0.04c 4.359 ± 0.03a SS含量 /
μg/g FW28.883 ± 0.31a 46.577 ± 0.27b 64.277 ± 0.19c 59.265 ± 0.19c Pro含量 /
μg/g FW22.794 ± 0.12a 19.750 ± 0.03a 35.925 ± 0.17b 40.482 ± 0.35b 叶绿素a /
mg/g FW0.430 ± 0.00a 0.449 ± 0.00ab 0.443 ± 0.00a 0.445 ± 0.00ab 叶绿素 b /
mg/g FW0.808 ± 0.00b 0.584 ± 0.02a 0.623 ± 0.02a 0.585 ± 0.02a 叶绿素总含量 /
mg/g FW1.238 ± 0.00b 1.033 ± 0.01a 1.066 ± 0.02a 1.030 ± 0.02a SOD活性 /
U/g FW419.542 ± 10.02a 431.441 ± 6.87ab 479.531 ± 2.48c 441.430 ± 5.05b POD活性 /
U/g FW11.725 ± 1.10a 15.535 ± 1.63a 25.331 ± 2.69b 27.879 ± 1.26b 注:同行不同小写字母表示处理间差异显著(P < 0.05)。 Note: Different lowercase letters indicate significant differences between treatments (P < 0.05). 表 2 低温胁迫及恢复后叶片单位横截面积比活性参数变化
Table 2 Changes in specific activity parameters per unit cross-sectional area of leaves after low temperature stress and recovery
处理
TreatmentABS/CSO DIO/CSO TRO/CSO ETO/CSO T1 5085.889 ± 126.80a 926.271 ± 34.89a 4159.618 ± 93.10ab 2367.616 ± 45.67c T2 5451.889 ± 72.15ab 1309.842 ± 96.39b 4142.047 ± 103.44ab 2297.065 ± 116.57c T3 5615.000 ± 96.28b 1383.823 ± 50.81b 4231.177 ± 77.20b 1937.618 ± 134.92b T4 5728.333 ± 266.44b 1833.883 ± 153.36c 3894.450 ± 131.31a 1291.324 ± 135.30a T1-H 5099.666 ± 126.60a 920.641 ± 33.09a 4239.601 ± 91.10b 2398.331 ± 44.77b T2-H 5317.111 ± 125.08a 1052.801 ± 38.45a 4264.310 ± 92.68b 2412.295 ± 100.46b T3-H 5429.833 ± 172.32a 1106.154 ± 56.32a 4323.679 ± 119.74b 2453.114 ± 84.32b T4-H 5513.555 ± 262.21a 2072.676 ± 465.71b 3440.880 ± 390.15a 1491.727 ± 270.88a 注:同列不同小写字母表示处理间差异显著(P < 0.05)。下同。 Note: Different lowercase letters in the same column indicate significant difference between treatments (P < 0.05). Same below. 表 3 低温胁迫及恢复后连翘幼苗叶片单位PSⅡ反应中心比活性参数变化
Table 3 Changes in specific activity parameters of unit PSII reaction center of Forsythia suspensa seedlings after cold stress and recovery
处理
TreatmentABS / RC TRO / RC DIO / RC ETO / RC T1 0.959 ± 0.04a 0.784 ± 0.03a 0.175 ± 0.01a 0.445 ± 0.01b T2 1.218 ± 0.06b 0.918 ± 0.03b 0.300 ± 0.04b 0.512 ± 0.03b T3 1.017 ± 0.03a 0.765 ± 0.02a 0.252 ± 0.02ab 0.346 ± 0.02a T4 1.275 ± 0.09b 0.863 ± 0.05ab 0.412 ± 0.04c 0.280 ± 0.03a T1-H 0.950 ± 0.04a 0.774 ± 0.03a 0.175 ± 0.01a 0.442 ± 0.01b T2-H 1.130 ± 0.03a 0.906 ± 0.02a 0.224 ± 0.01ab 0.512 ± 0.02b T3-H 1.037 ± 0.04a 0.826 ± 0.03a 0.211 ± 0.01ab 0.467 ± 0.01b T4-H 1.804 ± 0.41b 0.895 ± 0.07a 0.910 ± 0.42b 0.360 ± 0.03a 表 4 低温胁迫及恢复后连翘叶片供体侧和受体侧参数的变化
Table 4 Changes in donor side and receptor side parameters of Forsythia suspensa seedlings after cold stress and recovery
处理
TreatmentWk Sm N ψO φEO T1 0.259 ± 0.01a 15.663 ± 0.50a 12.160 ± 0.18a 0.571 ± 0.01c 0.467 ± 0.01c T2 0.310 ± 0.01ab 21.786 ± 1.47b 19.921 ± 1.43b 0.555 ± 0.02c 0.421 ± 0.02c T3 0.259 ± 0.00a 22.336 ± 3.11b 16.767 ± 1.97b 0.459 ± 0.03b 0.347 ± 0.03b T4 0.292 ± 0.01ab 12.877 ± 1.63a 10.897 ± 1.44a 0.333 ± 0.04a 0.230 ± 0.03a T1-H 0.258 ± 0.01a 15.363 ± 0.50ab 12.150 ± 0.18a 0.571 ± 0.01b 0.467 ± 0.01b T2-H 0.271 ± 0.01ab 16.596 ± 0.55bc 15.077 ± 0.77b 0.565 ± 0.01b 0.453 ± 0.01b T3-H 0.248 ± 0.01a 18.958 ± 0.89c 15.617 ± 0.82b 0.567 ± 0.01b 0.452 ± 0.01b T4-H 0.270 ± 0.01ab 12.948 ± 1.55a 11.173 ± 1.24a 0.435 ± 0.06a 0.286 ± 0.06a -
[1] Gao JP,Wallis JG,Jewell JB,Browse J. Trimethylguanosine synthase1 (TGS1) is essential for chilling tolerance[J]. Plant Physiol,2017,174 (3):1713−1727. doi: 10.1104/pp.17.00340
[2] 张晓虎. 洛南县连翘产地气候因素分析研究[J]. 陕西农业科学,2018,64(9):25−29. doi: 10.3969/j.issn.0488-5368.2018.09.009 [3] Wu DX,Kukkonen S,Luoranen J,Pulkkinen P,Repo T,et al. Influence of late autumn preconditioning temperature on frost hardiness of apple,blueberry and blackcurrant saplings[J]. Sci Hortic,2019,258:108755. doi: 10.1016/j.scienta.2019.108755
[4] Nejadsadeghi L,Maali-Amiri R,Zeinali H,Ramezanpour S,Sadeghzade B. Membrane fatty acid compositions and cold-induced responses in tetraploid and hexaploid wheats[J]. Mol Biol Rep,2015,42 (2):363−372. doi: 10.1007/s11033-014-3776-3
[5] Gill SS,Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants[J]. Plant Physiol Biochem,2010,48 (12):909−930. doi: 10.1016/j.plaphy.2010.08.016
[6] Yang QS,Wu JH,Li CY,Wei YR,Sheng O,et al. Quantitative proteomic analysis reveals that antioxidation mechanisms contribute to cold tolerance in plantain (Musa paradisiaca L.; ABB group) seedlings[J]. Mol Cell Proteomics,2012,11 (12):1853−1869. doi: 10.1074/mcp.M112.022079
[7] Xu J,Li Y,Sun J,Du L,Zhang Y. Comparative physiological and proteomic response to abrupt low temperature stress between two winter wheat cultivars differing in low temperature tolerance[J]. Plant Biol,2013,15 (2):292−303. doi: 10.1111/j.1438-8677.2012.00639.x
[8] Jin JJ,Zhang H,Zhang JF,Liu PP,Chen X,et al. Integrated transcriptomics and metabolomics analysis to characterize cold stress responses in Nicotiana tabacum[J]. BMC Genomics,2017,18 (1):496. doi: 10.1186/s12864-017-3871-7
[9] Gothandam KM,Nalini E,Karthikeyan S,Shin JS. OsPRP3,a flower specific proline-rich protein of rice,determines extracellular matrix structure of floral organs and its overexpression confers cold-tolerance[J]. Plant Mol Biol,2010,72 (1-2):125−135. doi: 10.1007/s11103-009-9557-z
[10] Yang Y,Yao N,Jia ZK,Jia J,Duan FJ,et al. Effect of exogenous abscisic acid on cold acclimation in two Magnolia species[J]. Biol Plant,2016,60 (3):555−562. doi: 10.1007/s10535-016-0623-5
[11] Thakur P,Kumar S,Malik JA,Berger JD,Nayyar H. Cold stress effects on reproductive development in grain crops:an overview[J]. Environ Exp Bot,2010,67 (3):429−443. doi: 10.1016/j.envexpbot.2009.09.004
[12] Ensminger I,Busch F,Huner NPA. Photostasis and cold acclimation:sensing low temperature through photosynthesis[J]. Physiol Plant,2006,126 (1):28−44. doi: 10.1111/j.1399-3054.2006.00627.x
[13] Kong FY,Deng YS,Zhou B,Wang GD,Wang Y,Meng QW. A chloroplast-targeted DnaJ protein contributes to maintenance of photosystem Ⅱ under chilling stress[J]. J Exp Bot,2014,65 (1):143−158. doi: 10.1093/jxb/ert357
[14] Wang ZY,Xia Q,Liu X,Liu WX,Huang WZ,et al. Phytochemistry,pharmacology,quality control and future research of Forsythia suspensa (Thunb.) Vahl:a review[J]. J Ethnopharmacol,2018,210:318−339. doi: 10.1016/j.jep.2017.08.040
[15] Li C,Wei Q,Zou ZH,Sun CZ,Wang Q,et al. A lignan and a lignan derivative from the fruit of Forsythia suspensa[J]. Phytochem Lett,2019,32:115−118. doi: 10.1016/j.phytol.2019.05.009
[16] Heath RL,Packer L. Photoperoxidation in isolated chloroplast:Ⅰ. Kinetics and stoichiometry of fatty acid peroxidation[J]. Arch Biochem Biophys,1968,125 (1):189−198. doi: 10.1016/0003-9861(68)90654-1
[17] 王永军,杨今胜,袁翠平,柳京国,李登海,董树亭. 超高产夏玉米花粒期不同部位叶片衰老与抗氧化酶特性[J]. 作物学报,2013,39(12):2183−2191. Wang YJ,Yang JS,Yuan CP,Liu JG,Li DH,Dong ST. Characteristics of senescence and antioxidant enzyme activities in leaves at different plant parts of summer maize with the super-high yielding potential after anthesis[J]. Acta Agronomica Sinica,2013,39 (12):2183−2191. doi: 10.3724/SP.J.1006.2013.02183 Wang YJ, Yang JS, Yuan CP, Liu JG, Li DH, Dong ST. Characteristics of senescence and antioxidant enzyme activities in leaves at different plant parts of summer maize with the super-high yielding potential after anthesis[J]. Acta Agronomica Sinica, 2013, 39(12): 2183-2191. doi: 10.3724/SP.J.1006.2013.02183
[18] Redmann RE,Haraldson J,Gusta LV. Leakage of UV-absorbing substances as a measure of salt injury in leaf tissue of woody species[J]. Physiol Plant,1986,67 (1):87−91. doi: 10.1111/j.1399-3054.1986.tb01267.x
[19] Lichtenthaler HK,Wellburn AR. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents[J]. Biochem Soc Trans,1983,11 (5):591−592. doi: 10.1042/bst0110591
[20] Rosa M,Hilal M,González JA,Prado FE. Low-temperature effect on enzyme activities involved in sucrose-starch partitioning in salt-stressed and salt-acclimated cotyledons of quinoa (Chenopodium quinoa Willd.) seedlings[J]. Plant Physiol Biochem,2009,47 (4):300−307. doi: 10.1016/j.plaphy.2008.12.001
[21] Bates LS,Waldren RP,Teare ID. Rapid determination of free proline for water-stress studies[J]. Plant Soil,1973,39 (1):205−207. doi: 10.1007/BF00018060
[22] Haldimann P,Strasser RJ. Effects of anaerobiosis as probed by the polyphasic chlorophyll a fluorescence rise kinetic in pea (Pisum sativum L. )[J]. Photosyn Res,1999,62 (1):67−83. doi: 10.1023/A:1006321126009
[23] Strasser RJ,Tsimilli-Michael M,Qiang S,Goltsev V. Simultaneous in vivo recording of prompt and delayed fluorescence and 820-nm reflection changes during drying and after rehydration of the resurrection plant Haberlea rhodopensis[J]. Biochim Biophys Acta Bioenerg,2010,1797 (6-7):1313−1326. doi: 10.1016/j.bbabio.2010.03.008
[24] Arslan Ö,Eyidoğan F,Ekmekçi Y. Freezing tolerance of chickpea:biochemical and molecular changes at vegetative stage[J]. Biol Plant,2018,62 (1):140−148. doi: 10.1007/s10535-017-0760-5
[25] Lukatkin AS,Brazaitytė A,Bobinas C,Duchovskis P. Chilling injury in chilling-sensitive plants:a review[J]. Zemdirbyste,2012,99 (2):111−124.
[26] 程祥飞,王磊,聂林杰,李永华. 低温胁迫下菊花叶片叶绿素荧光特性与抗氧化酶活性的变化[J]. 河南农业科学,2018,47(4):104−108. Cheng XF,Wang L,Nie LJ,Li YH. Chlorophyll fluorescence characteristics and antioxidant enzyme activities of chrysanthemum leaves under low temperature stress[J]. Journal of Henan Agricultural Sciences,2018,47 (4):104−108. Cheng XF, Wang L, Nie LJ, Li YH. Chlorophyll fluorescence characteristics and antioxidant enzyme activities of chrysanthemum leaves under low temperature stress[J]. Journal of Henan Agricultural Sciences, 2018, 47(4): 104-108.
[27] Fu ZZ,Lei YK,Peng DD,Li Y. Population genetics of the widespread shrub Forsythia suspensa (Oleaceae) in warm-temperate China using microsatellite loci:implication for conservation[J]. Plant Syst Evol,2016,302 (1):1−9. doi: 10.1007/s00606-015-1241-y
[28] 李清亚,路斌,赵佳伟,栗浩,李艳,等. 不同豆梨品种对低温胁迫的生理响应及抗寒性评价[J]. 西北农林科技大学学报(自然科学版),2020,48(1):86−94,110. Li QY,Lu B,Zhao JW,Li H,Li Y,et al. Physiological response and cold resistance evaluation of different Pyrus calleryan varieties under low temperature stress[J]. Journal of Northwest A & F University (Natural Science Edition) ,2020,48 (1):86−94,110. Li QY, Lu B, Zhao JW, Li H, Li Y, et al. Physiological response and cold resistance evaluation of different Pyrus calleryan varieties under low temperature stress[J]. Journal of Northwest A & F University (Natural Science Edition), 2020, 48(1): 86-94, 110.
[29] Zhang Q,Liu YX,Yu QQ,Ma Y,Gu WR,Yang DG. Physiological changes associated with enhanced cold resistance during maize (Zea mays) germination and seedling growth in response to exogenous calcium[J]. Crop Pasture Sci,2020,71 (6):529−538. doi: 10.1071/CP19510
[30] Feng Q,Yang S,Wang YJ,Lu L,Sun MT,et al. Physiological and molecular mechanisms of ABA and CaCl2 regulating chilling tolerance of cucumber seedlings[J]. Plants,2021,10 (12):2746. doi: 10.3390/plants10122746
[31] Ignatenko A,Talanova V,Repkina N,Titov A. Exogenous salicylic acid treatment induces cold tolerance in wheat through promotion of antioxidant enzyme activity and proline accumulation[J]. Acta Physiol Plant,2019,41 (6):80. doi: 10.1007/s11738-019-2872-3
[32] Wang SQ,Zhao HH,Zhao LM,Gu CM,Na YG,Scholes GD. Application of brassinolide alleviates cold stress at the booting stage of rice[J]. J Integr Agric,2020,19 (4):975−987. doi: 10.1016/S2095-3119(19)62639-0
[33] Mirkovic T,Ostroumov EE,Anna JM,van Grondelle R,Govindjee,et al. Light absorption and energy transfer in the antenna complexes of photosynthetic organisms[J]. Chem Rev,2017,117 (2):249−293. doi: 10.1021/acs.chemrev.6b00002
[34] Borawska-Jarmułowicz B,Mastalerczuk G,Kalaji HM,Carpentier R,Pietkiewicz S,Allakhverdiev SI. Photosynthetic efficiency and survival of Dactylis glomerata and Lolium perenne following low temperature stress[J]. Russ J Plant Physiol,2014,61 (3):281−288. doi: 10.1134/S1021443714030029
[35] Gururani MA,Venkatesh J,Tran LSP. Regulation of photosynthesis during abiotic stress-induced photoinhibition[J]. Mol Plant,2015,8 (9):1304−1320. doi: 10.1016/j.molp.2015.05.005
[36] Guisse B,Srivastava A,Strasser R. The polyphasic rise of the chlorophyll a fluorescence (O-K-J-I-P) in heat-stressed leaves[J]. Arch Sci,1995,48 (2):147−160.
[37] Kalaji HM,Jajoo A,Oukarroum A,Brestic M,Zivcak M,et al. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions[J]. Acta Physiol Plant,2016,38 (4):102. doi: 10.1007/s11738-016-2113-y
[38] Murchie EH,Lawson T. Chlorophyll fluorescence analysis:a guide to good practice and understanding some new applications[J]. J Exp Bot,2013,64 (13):3983−3998. doi: 10.1093/jxb/ert208
[39] Zhou R,Wu Z,Wang X,Rosenqvist E,Wang YL,et al. Evaluation of temperature stress tolerance in cultivated and wild tomatoes using photosynthesis and chlorophyll fluorescence[J]. Hortic Environ Biotechnol,2018,59 (4):499−509. doi: 10.1007/s13580-018-0050-y
[40] Lee HY,Hong YN,Chow WS. Photoinactivation of photosystem Ⅱ complexes and photoprotection by non-functional neighbours in Capsicum annuum L. leaves[J]. Planta,2001,212 (3):332−342. doi: 10.1007/s004250000398
[41] Guha A,Sengupta D,Reddy AR. Polyphasic chlorophyll a fluorescence kinetics and leaf protein analyses to track dynamics of photosynthetic performance in mulberry during progressive drought[J]. J Photochem Photobiol B,2013,119:71−83. doi: 10.1016/j.jphotobiol.2012.12.006
[42] 曲丽娜,许楠,张会慧. 风箱果和紫叶风箱果叶片光系统Ⅱ功能对秋季低温的响应[J]. 东北林业大学学报,2018,46(8):42−48. Qu LN,Xu N,Zhang HH. Response of photosynthetic function of photosystem Ⅱ in leaves of Physocarpus amurensis maxim and P. opulifolius in autumn low temperature stress in cold regions[J]. Journal of Northeast Forestry University,2018,46 (8):42−48. Qu LN, Xu N, Zhang HH. Response of photosynthetic function of photosystem II in leaves of Physocarpus amurensis maxim and P. opulifolius in autumn low temperature stress in cold regions[J]. Journal of Northeast Forestry University, 2018, 46(8): 42-48.
[43] Maliba BG,Inbaraj PM,Berner JM. The effect of ozone and drought on the photosynthetic performance of canola[J]. J Integr Agric,2018,17 (5):1137−1144. doi: 10.1016/S2095-3119(17)61834-3
[44] Roháček K. Chlorophyll fluorescence parameters:the definitions,photosynthetic meaning,and mutual relationships[J]. Photosynthetica,2002,40 (1):13−29. doi: 10.1023/A:1020125719386
[45] Snider JL,Thangthong N,Pilon C,Virk G,Tishchenko V. OJIP-fluorescence parameters as rapid indicators of cotton (Gossypium hirsutum L. ) seedling vigor under contrasting growth temperature regimes[J]. Plant Physiol Biochem,2018,132:249−257. doi: 10.1016/j.plaphy.2018.09.015
-
期刊类型引用(8)
1. 陈嫣,王志东,王晓东,段瑞. 临汾地区连翘繁育栽培技术及抚育管理. 特种经济动植物. 2024(01): 91-93 .  百度学术
百度学术
2. 郝丽叶,路琦,阿力木·阿木提,张锐,徐崇志. 低温胁迫对扁桃优株生理特性的影响. 山西农业科学. 2024(01): 68-78 .  百度学术
百度学术
3. 陶文楷,郝明灼,刘佳琪,邹义萍,梁有旺. 金叶斯蒂芬冬青与原种斯蒂芬冬青耐盐性比较分析. 北方园艺. 2024(11): 49-55 .  百度学术
百度学术
4. 冯朝晖,郑婷婷,田瑞昌,牛燕燕. 倒春寒对王屋山区连翘的影响及防控措施. 西北园艺. 2024(09): 29-31 .  百度学术
百度学术
5. 巩永杰,田海燕,魏家萍,崔俊美,武泽峰,董小云,郑国强,王莹,王小霞,刘自刚. 冬播对冬性/春性甘蓝型油菜萌发与花期生理生化的影响. 华北农学报. 2024(06): 106-114 .  百度学术
百度学术
6. 马彪,南丽丽,汪堃,姚宇恒,陈洁,夏静. 30份苜蓿种质材料对低温胁迫的生理响应及其耐寒性评价. 草原与草坪. 2024(06): 170-180 .  百度学术
百度学术
7. 金哲,董环宇,金铭路,任昭辉,苏亮,李俊明,朴世领. 外施水杨酸诱导烟草苗期对低温胁迫耐寒性的最适浓度筛选. 延边大学农学学报. 2023(02): 14-20 .  百度学术
百度学术
8. 郑锋,白小明,冉福,李娟霞,康瑞卿,董凯,郑四海. 10份沿阶草族种质对低温胁迫的生理响应及抗寒性评价. 中国草地学报. 2023(12): 21-32 .  百度学术
百度学术
其他类型引用(17)
-
其他相关附件
-
PDF格式
李永 - 附表 点击下载(201KB)
-



 下载:
下载: